ตำรามาตรฐานยาสมุนไพรไทย
Thai Herbal Pharmacopoeia
สำนักยาและวัตถุเสพติด กรมวิทยาศาสตร์การแพทย์ กระทรวงสาธารณสุข
Bureau of Drug and Narcotic, Department of Medical Sciences, Ministry of Public Health(Tinospora crispa (L.) Hook.f. & Thomson)
(Nelumbo nucifera Gaertn.)
(Centella asiatica (L.) Urb.)
(Centella Dry Extract)
(Centella Cream)
(Mesua ferrea L.)
(Piper sarmentosum Roxb.)
(Piper sarmentosum Roxb.)
(Pterocarpus santalinus L. f.)
(Santalum album L.)
(Senna tora (L.) Roxb.)
(Senna alata (L.) Roxb.)
(Senna Alata Tea)
(Piper retrofractum Vahl)
(Myristica fragrans Houtt)
(Andrographis paniculata (Burm. f.) Nees)
(Andrographis Capsules)
(Allium ascalonicum L.)
(Ocimum tenuiflorum L.)
(Curcuma longa L.)
(Turmeric Capsules)
(Turmeric Dry Extract)
(Turmeric Dry Extract Capsules)
(Arcangelisia flava (L.) Merr.)
(Curcuma sp.)
Harrisonia perforata (Blanco) Merr.
(Aristolochia pierrei Lecomte)
(Zingiber officinale Roscoe)
(Ginger Capsules)
(Ginger Tea)
(Cassia fistula L.)
(Nardostachys jatamansi (D. Don) DC.)
(Angelica sinensis (Oliv.) Diels)
Artemisia annua L.
(Ligusticum sinense Oliv. cv. Chuanxiong)
(Neopicrorhiza scrophulariiflora Pennell)
(Atractylodes lancea (Thunb.) DC.)
(Aucklandia lappa Decne)
(Terminalia chebula Retz.)
(Angelica dahurica (Hoffm.) Benth. & Hook. f. ex Franch. & Sav. var. dahurica)
(Kaempferia parviflora Wall. ex Baker)
(Hibiscus sabdariffa L.)
(Roselle Tea)
(Allium sativum L.)
(Zingiber zerumbet (L.) Sm.)
(Wurfbainia testacea (Ridl.) Škorničk.& A. D. Poulsen)
(Cannabis sativa L.)
(Myristica fragrans Houtt)
(Dracaena cochinchinensis (Lour.) S. C. Chen)
(Ficus racemosa L.)
(Hyptis suaveolens (L.) Poit.)
Clerodendrum indicum (L.) Kuntze
(Phyllanthus emblica L.)
(Citrus hystrix DC.)
(Citrus hystrix DC.)
(Areca catechu L.)
(Momordica charantia L.)
Moringa oleifera Lam.
(Aegle marmelos (L.) Corrêa)
(Solanum trilobatum L.)
(Morus alba L.)
Gynostemma pentaphyllum(Thunb.)
Makino
(Clinacanthus nutans (Burm. f.) Lindau)
(Cissus quadrangularis L.)
(Mimusops elengi L.)
(Zingiber montanum (J. König) Link. ex A. Dietr.)
(Piper betle L.)
(Capsicum annuum L.)
(Capsicum Oleoresin)
(Capsicum Gel)
(Piper nigrum L.)
(Piper nigrum L.)
(Eurycoma longifolia Jack)
(Thunbergia laurifolia Lindl.)
(Piper wallichii (Miq.) Hand.-Mazz.)
Senna garrettiana (Craib) H. S. Irwin & Barneby
(Terminalia bellirica (Gaertn.) Roxb.)
(Terminalia chebula Retz.)
(Caesalpinia bonduc (L.) H. Roxb.)
(Tarlmounia elliptica (DC.) H. Rob., S. C. Keeley, Skvaria & R. Chan)
(Hog Creeper Vine Dry Extract Capsiles)
(Hog Creeper Vine Dry Extract)
(Brachypterum scandens (Roxb.) Miq.)
(Lepidium sativum L.)
(Nigella sativa L.)
(Cuminum cyminum L.)
(Foeniculum vulgare Mill.)
(Plantago ovata Forssk.)
(Pimpinella anisum L.)
(Carum carvi L.)
(Anethum graveolens L.)
(Trachyspermum ammi (L.) Sprague)
Albizia procera (Roxb.) Benth.
(Acorus calamus L.)
(Tiliacora triandra (Colebr.) Diels)
Cyanthillium cinereum (L.) H. Rob.
(Orthosiphon aristatus (Blume) Miq.)
Murdannia loriformis (Hassk.) R. S. Rao & Kammathy
(Capparis micracantha DC.)
(Chrysopogon zizanioides (L.) Roberty)
(Cyperus rotundus L.)
(Cannabis sativa L.)
(Syzygium aromaticum (L.) Merr. & L. M. Perry)
(Boesenbergia rotunda (L.) Mansf.)
(Acanthus ebracteatus Vahl)
(Acanthus ilicifolius L.)
(Kaempferia galanga L.)
(Curcuma comosa Roxb.)
Betula alnoides Buch.-Ham. ex D. Don
Cannabis sativa L.
Carthamus tinctorius L
Mitragyna speciosa (Korth.) Havil
Mallotus repandus (Rottler) Müll. Arg
Azadirachta indica A. Juss. var. siamensis Valeton
Azadirachta indica A. Juss. var. siamensis Valeton
Punica granatum L.
Rhinacanthus nasutus (L.) Kurz
Baliospermum solanifolium (Burm.) Suresh
Curcuma aeruginosa Roxb
Boesenbergia kingii Mood & L. M. Prince
Senegalia rugata (Lam.) Britton & Rose
Acacia concinna (Willd.) DC.
Senegalia rugata (Lam.) Britton & Rose
Acacia concinna (Willd.) DC.
Senna alexandriana Mill. var. alexandriana
Cassia acutifolia Delile, Cassia angustifolia Vahl
Butea superba Roxb. ex Willd.
[Plaso superba (Roxb. ex Willd.) Kuntze, Rudolphia superba (Roxb. ex Willd.) Poir.
Pueraria candollei Graham
ex Benth. var. mirifica (Airy Shaw & Suvat.) Niyomdham
Streblus asper Lour.
Suregada multiflora (A. Juss.) Baill. (Gelonium
multiflorum A. Juss.
Plumbago zeylanica L.
Plumbago indica L.
Biancaea sappan (L.) Tod.
Ziziphus attopensis Pierre
Streblus asper Lour.
Justicia gendarussa Burm. f.
Enhalus acoroides (L. f.) Royle
Bridelia ovata Decne.
Tamarindus indica L.
Citrus × aurantiifolia (Christm.) Swingle
Garcinia mangostana L.
Blumea balsamifera (L.) DC
Persicaria odorata (Lour.) Soják
Zingiber montanum (J. König) Link ex A. Dietr.
Mammea siamensis (Miq.) T. Anderson
Citrus maxima (Burm.) Merr.
Citrus × aurantium L. ‘Som Sa'
Punica granatum L.
Rhinacanthus nasutus (L.) Kurz
Andrographis Herb is the dried aerial part of Andrographis paniculata (Burm. f.) Nees (Justicia paniculata Burm. f.) (Family Acanthaceae), Herbarium Specimen Number: DMSC 821, 854, Crude Drug Number: DMSc 0407.
Constituents Andrographis Herb contains a large quantity of bitter diterpenoid lactones which mainly are andrographolide, neoandrographolide, 14-deoxy-11,12-didehydroandrographolide (dehydroandrographolide), dideoxyandrographolide (andrograpanin), etc. and their glycosides. It also contains β-sitosterol-D-glucoside, flavonoids, etc.
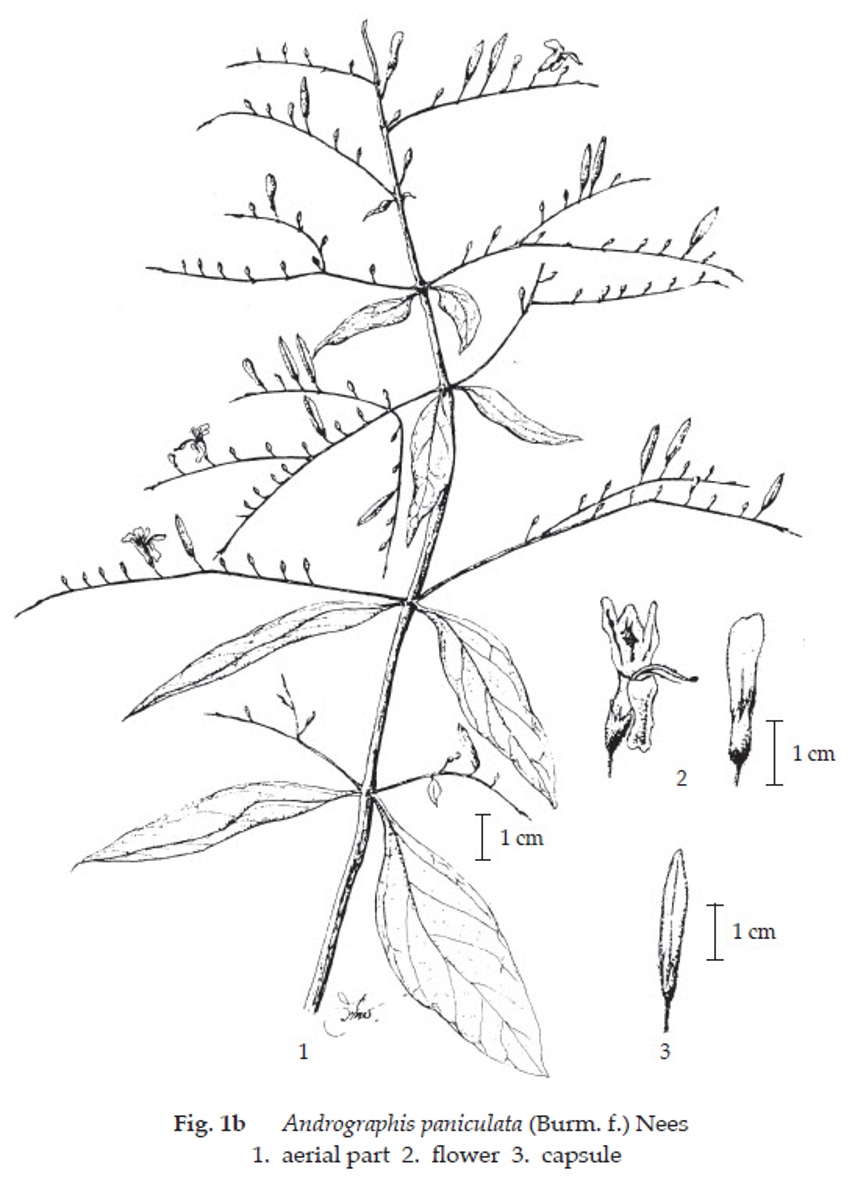
Description of the plant (Figs. 1a, 1b) Annual herb up to 1 m tall; stem, erect, acutely quadrangular. Leaves simple, opposite, lanceolate, 2 to 12 cm long, 1 to 3 cm wide, acute at both ends, glabrous, entire-slightly undulate, upper ones often bractiform; petiole short. Inflorescence patent, terminal and axillary, in panicle, 10 to 30 cm long; bract small, pedicel short. Flower: calyx 5-partite, small, linear; corolla tube narrow, about 6 mm long, limb not shorter than the tube, bilabiate, upper lip oblong, white with a yellowish top, lower lip broadly cuneate, trifid, white with violet marking; stamens 2, inserted in the throat and far exserted, anther basally bearded; ovary superior, 2-celled, s tyle far exserted. Fruit capsule, erect, linear-oblong, 1 to 2 cm long, 2 to 5 mm wide, compressed, longitudinally furrowed on the broad faces, thinly glandular-hairy. Seed small, subquadrate.
Description Odour, slight and specific; taste, extremely bitter.
Macroscopical (Fig. 1a) Dried herb occurs as a mixture of broken, crisp, mainly dark green lanceolate leaf and quadrangularly stem; capsule fruit and small flower occasionally found.
Microscopical (Figs. 2a, 2b, 2c, 2d) Surface and transverse sections of the leaf through lamina and midrib region show the following characters: upper epidermis, a layer of cells; stomata absent; covering trichomes, glandular, unicellular and multicellular present; cicatrices rarely seen; lithocysts, fairly large, 27 μm × 96 μm to 30 μm × 210 μm in size and 49 μm thick. Palisade cells, columnar. Collenchyma occurs in the midrib, beneath upper and lower epidermises. Spongy cells, parenchymatous. Vascular bundles, composed of lignified xylem in the upper part and phloem in the lower part. Vessels, spiral, scalariform and reticulate. Lower epidermis, a layer of wavy-walled cells; stomata, diacytic; covering trichomes and lithocysts present.
Transverse section of the stem shows the following characters: epidermis, a layer of cells; stomata, diacytic; covering trichomes and lithocys ts present. Collenchyma, densely found at the corners of stem. Parenchyma, containing chlorophyll. Endodermis, a layer of thick-walled cells. Vessels, spiral, scalariform and pitted. Pith, large parenchyma cells.


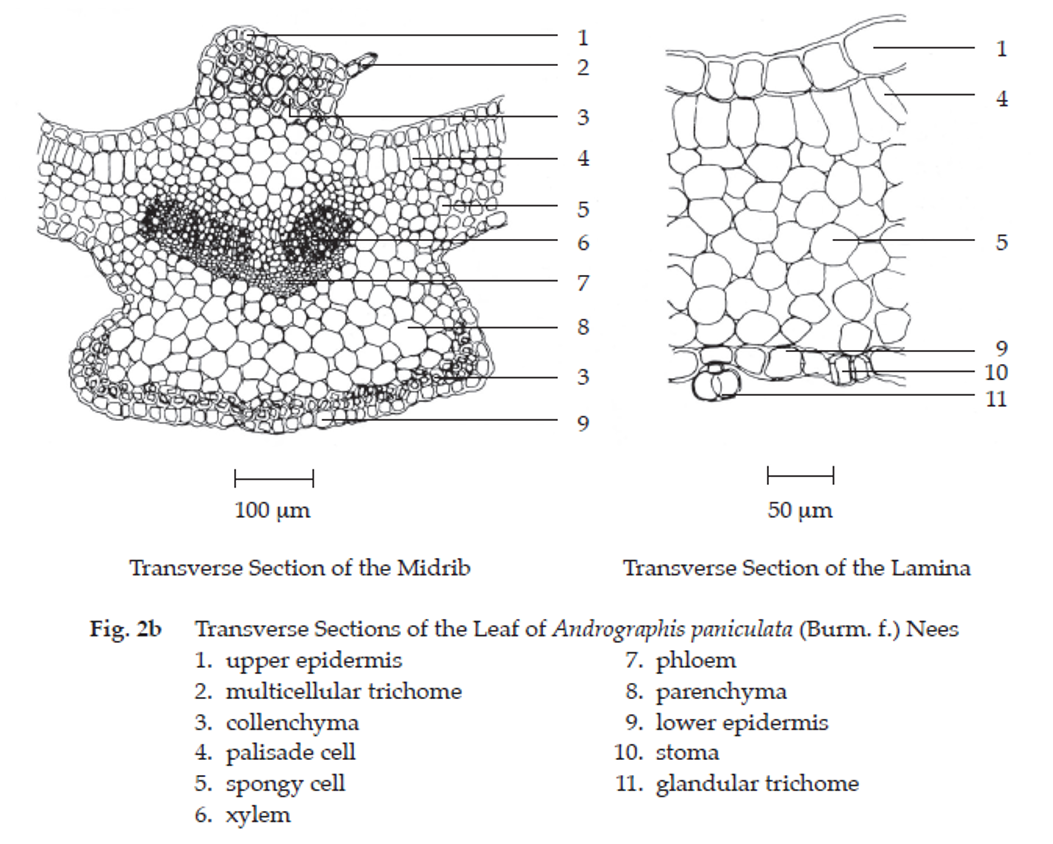
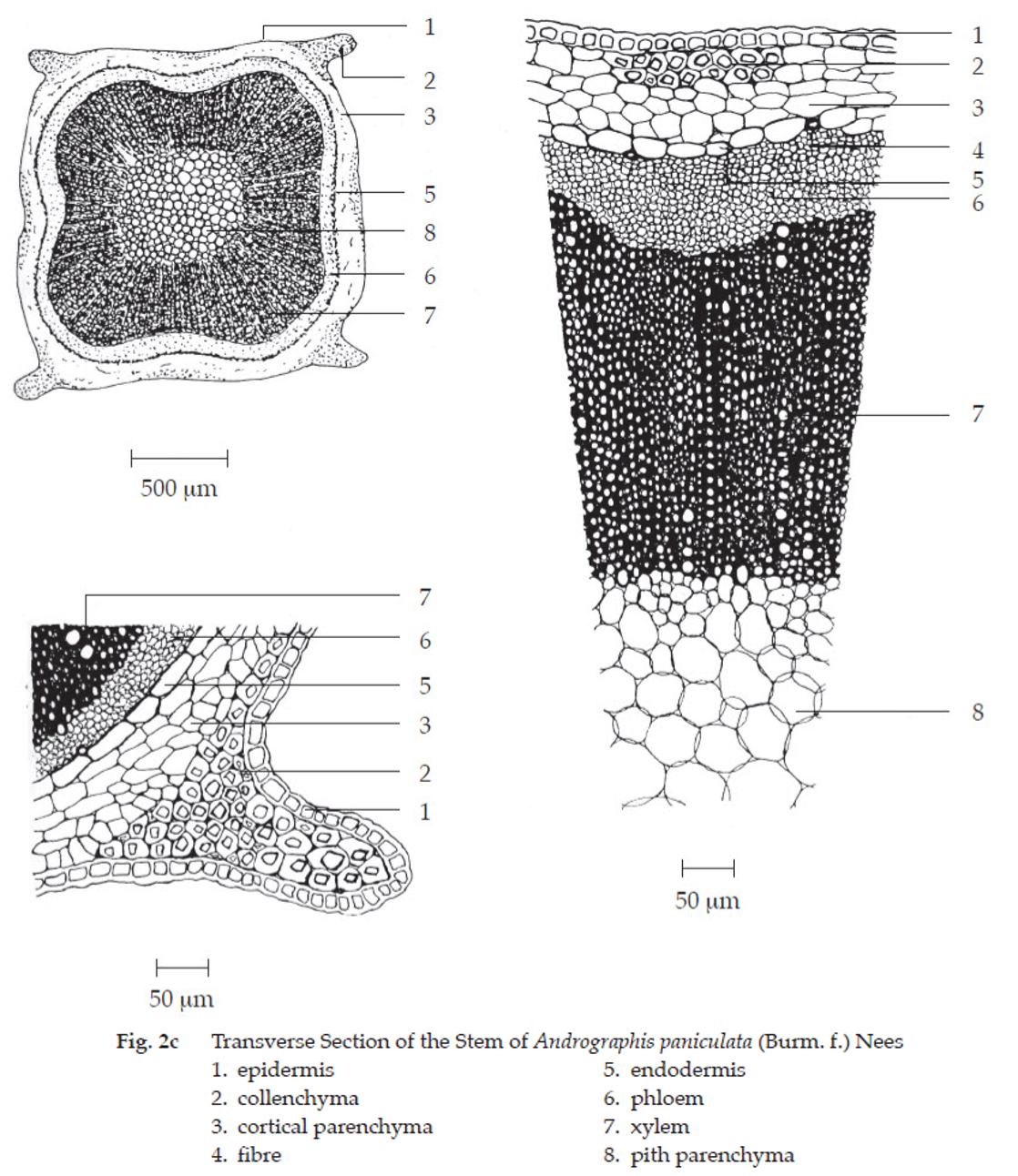

Andrographis Herb in powder possesses the diagnostic microscopical characters of the unground drug
Packaging and storage Andrographis Herb shall be kept in well-closed containers, protected from light, and stored in a dry place. It should be used within 1 year and air-dried every 2 to 3 months.
Identification
A. To about 1 g of the sample, in powder, add 20 mL of ethanol, boil in a water-bath for 5 minutes. To the filtrate, add 300 mg of decolorizing charcoal, stir and filter (solution 1). To 1 mL of solution 1, add 2 drops of a 2 per cent w/v solution of 3,5-dinitrobenzoic acid in methanol and 2 drops of a 5.7 per cent w/v solution of potassium hydroxide in methanol: a purplish red colour develops.
B. To 1 mL of solution 1, add several drops of a 5.7 per cent w/v solution of potassium hydroxide in methanol until it shows a red colour. Set aside for 10 to 15 minutes: the colour is changed to yellow.
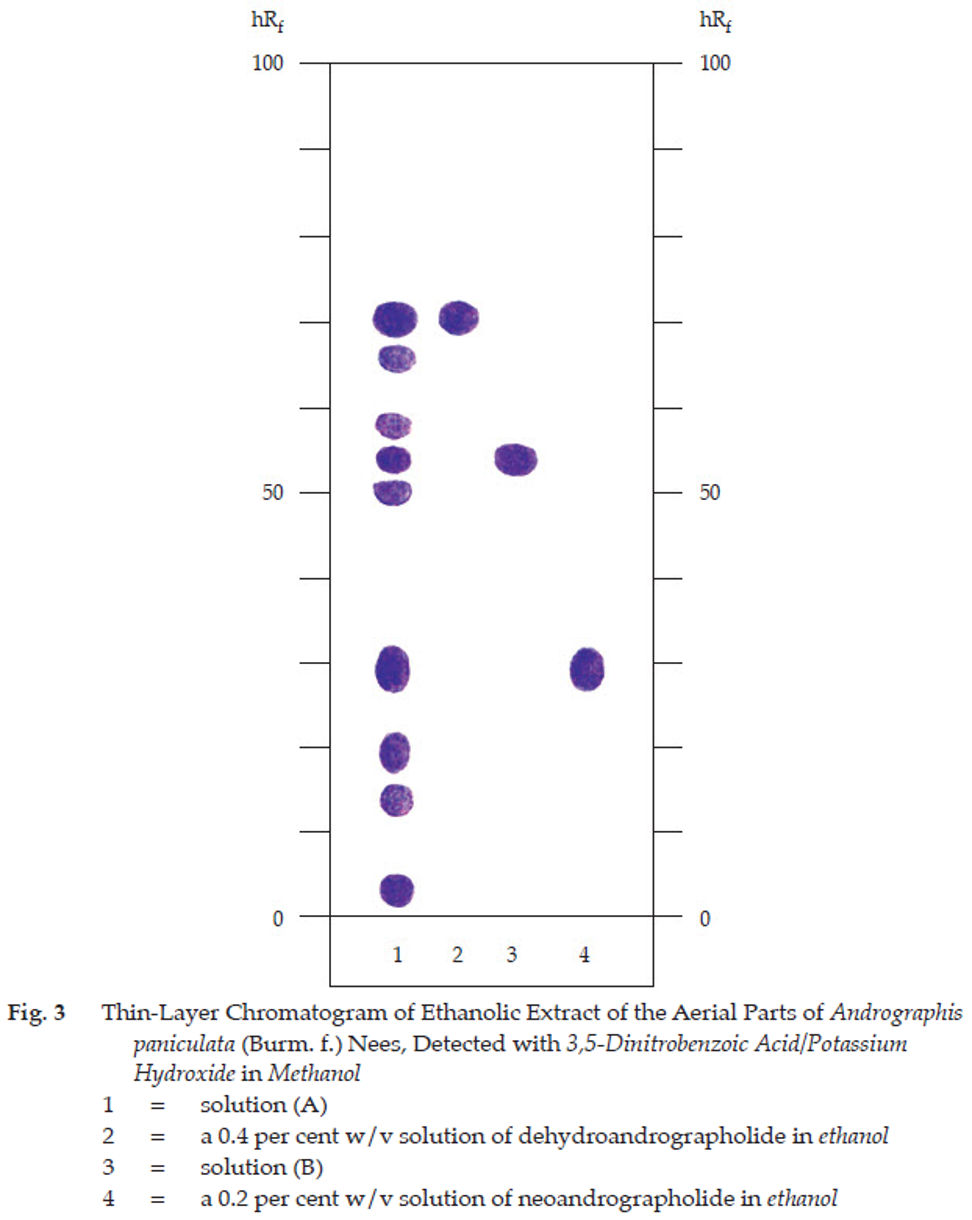
C. Carry out the test as described in the “Thin-Layer Chromatography” (Appendix 3.1), using silica gel GF254 as the coating substance and a mixture of 85 volumes of chloroform and 15 volumes of absolute ethanol as the mobile phase. Apply separately to the plate, 5 μL each of the following solutions. Prepare solution (A) by boiling 1 g of the sample, in powder, with 20 mL of ethanol on a water-bath for 5 minutes and filter. To the filtrate, adding 300 mg of decolorizing charcoal, stirring, and filtering. Evaporate the filtrate under reduced pressure to dryness, and dissolve the residue in 1 mL of warm ethanol (80 per cent). For solution (B), dissolve 2 mg of andrographolide in 1 ml of ethanol. After removal of the plate, allow it to dry in air and examine under ultraviolet light (254 nm), marking the quenching spots. The chromatogram obtained with solution (A) shows a quenching spot (hRf values 52 to 56) corresponding to the andrographolide spots from solution (B) and other four spots of different hRf values (Table 1); see also Fig. 3. Spray the plate with a 2 per cent w/v solution of 3,5-dinitrobenzoic acid in methanol and then with an excess of a 5.7 per cent w/v solution of potassium hydroxide in methanol; the spot due to andrographolide is dark violet. Two dark violet spots due to the spot numbers 4 and 9 in Table 1 and other violet and dark violet spots are also observed (Table 1); see also Fig. 3.
Table 1 hRf Values of Components in Ethanolic Extract of the Aerial Parts of Andrographis paniculata (Burm. f.) Nees
| Spot | hRf Value | Detection | |
| UV 254 | 3,5-Dinitrobenzoic Acid/ Potassium Hydroxide in Methanol |
||
| 1 | 1-5 | - | dark violet |
| 2 | 11-15 | quenching | violet |
| 3 | 18-22 | quenching | violet |
| 4* | 28-32 | - | dark violet |
| 5 | 49-51 | - | violet |
| 6** | 52-56 | quenching | dark violet |
| 7 | 57-59 | quenching | violet |
| 8 | 66-68 | - | violet |
| 9*** | 69-71 | quenching | dark violet |
*neoandrographolide
**andrographolide
***dehydroandrographolide
Loss on drying Not more than 11.0 per cent w/w after drying at 105° to constant weight (Appendix 4.15).
Foreign matter Not more than 2.0 per cent w/w (Appendix 7.2).
Acid-insoluble ash Not more than 2.0 per cent w/w (Appendix 7.6).
Ethanol (85 per cent)-soluble extractive Not less than 13.0 per cent w/w (Appendix 7.12).
Water-soluble extractive Not less than 18.0 per cent w/w (Appendix 7.12).
Total lactones content Not less than 6.0 per cent w/w of total lactones, calculated as andrographolide, when determined by the following method.
Procedure Place about 1 g of Andrographis Herb in fine powder, accurately weighed, in a 100-mL round-bottomed flask, add 50 mL of ethanol (85 per cent), reflux in a water-bath for 2 hours, and filter. Wash the marc with sufficient amount of ethanol (85 per cent) until the last washing is almost colourless. Combine the washings and the filtrate and allow to cool. Add 1 mL of basic lead acetate TS, set aside for 15 minutes, filter, and wash the precipitate with ethanol until the last washing is no longer green. Combine the washings and the filtrate, add dropwise with swirling 1 mL of a 25 per cent w/v solution of sodium sulfate and mix well. Set aside for 1 hour, add 500 mg of decolorizing charcoal, and reflux in a water-bath for 10 minutes. Filter through the Büchner funnel containing 500 mg of decolorizing charcoal and wash with three 2-mL portions of hot ethanol. Combine the washings and the filtrate, add 20 mL of distilled water, allow to cool, and neutralize with 0.1 M sodium hydroxide, using phenolphthalein TS as indicator. Add 5.0 mL of 0.1 M sodium hydroxide VS, reflux in a water-bath for 30 minutes, allow to cool, and titrate with 0.05 M hydrochloric acid VS. Perform a blank determination (Residual Titrations, Appendix 6.17). Each ml of 0.1 M sodium hydroxide VS is equivalent to 35.05 mg of total lactones calculated as andrographolide (C20H30O5).
Andrographolide content Not less than 1.0 per cent w/w of andrographolide (C20H30O5). Carry out the determination as described in the “Liquid Chromatography” (Appendix 3.5).
Mobile phase Prepare a mixture of 52 volumes of distilled water and 48 volumes of methanol. Make adjustments if necessary.
Standard preparations Dissolve an accurately weighed quantity of andrographolide in sufficient methanol and dilute with Mobile phase to obtain a stock solution having a known concentration of about 200 μg per mL. Dilute this solution quantitatively, and stepwise with Mobile phase to obtain six solutions having known concentrations of 20, 40, 60, 80, 100, and 140 μg per mL.
Sample preparation Reflux about 400 mg of Andrographis Herb, in fine powder, accurately weighed, with 50 mL of a mixture of equal volumes of dichloromethane and methanol in a water-bath for 30 minutes. Filter and evaporate the filtrate at 50° under reduced pressure to dryness. Dissolve the residue in sufficient methanol, transfer quantitatively to a 100-mL volumetric flask, dilute with Mobile phase to volume, and mix. Filter through a nylon membrane having a 0.45-μm porosity.
Chromatographic system The chromatographic procedure may be carried out using (a) a stainless steel column (15 cm × 3.9 mm) packed with octadecylsilane chemically bonded to porous silica or ceramic microparticles (5 μm), (b) a guard column (20 mm × 3.9 mm) packed with octadecylsilane chemically bonded to porous silica or ceramic microparticles (5 μm), (c) Mobile phase at a flow rate of about 1 mL per minute, and (d) an ultraviolet photometer set at 224 nm.
To determine the suitability of the chromatopraphic system, chromatograph Standard preparation having a known concentration of 60 μg per mL and record the peak responses as directed under Procedure and Calculation: the symmetry factor for the andrographolide peak is not more than 2.0 and the relative standard deviation for replicate injections is not more than 2.0 per cent.
Procedure and Calculation Separately inject about 20 μL each of Standard preparations into the chromatograph, record the chromatograms and measure the responses for the major peaks. Plot the readings and draw the standard curve of best fit. Inject about 20 μL of Sample preparation into the chromatograph, record the chromatogram and measure the response for the major peak. By reference to the standard curve, calculate the content of andrographolide (C20H30O5) in the Andrographis Herb taken.
Dose 0.5 to 2 g four times a day after meals and at bedtime.